|
vinorelbine tartrate
vinorelbine tartrate
|
Item |
Standard |
|
Appearance |
white powder |
|
Solubility |
freely soluble in water |
|
Differentiation |
IR |
Sample's infrared spectrum conforms to the reference standard. |
|
HPLC |
The retention time of the main ingredient in the chromatogram conforms to the reference standard. |
|
Tartaric acid reaction |
Tartaric acid: positive |
|
Clarity of Solution |
The solution is clarifying( 100mg/10ml water) |
|
Solution Color |
Absorbance ≤0.03 ( 420nm , 1% aqueous solution) |
|
Moisture |
≤ 4.0% |
|
Residue on Ignition |
≤ 0.1% |
|
PH |
3.3-3.80 ( 10mg/ml ) |
|
Related Substance |
Photolytic product yield |
≤ 0.3% |
|
Single impurity |
≤ 0.2% |
|
Total impurities |
≤ 0.7% |
|
Assaying |
98.0~102.0% ( on dried base ) |
|
Purity |
≥ 99% (HPLC) |
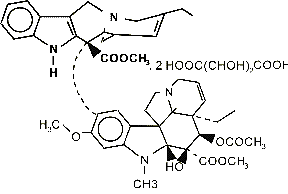
【Drug Name】Vinorelbine Ditartrate
【Chemical Name】3',4'-Didehydro-4'-deoxy-C'-norvincaleukoblastine [R-(R*,R*)-2-3-dihydroxybutanedioate (1:2)salt]
【Structural Formula】C45H54N4O8?C4H6O6
【Molecular Weight】1079.12
【Properties】This product is white or white like powder, easy to dissolve in water.
【Adaptive Disease】This product is broad-spectrum antineoplastic, is the vinblastine's semisynthetic derviation, mainly used for many kinds of tumor diseases.
【Usage 】Administration only by vena.
【Does Type】Injection
【Quality Standard】BP98 / USP24。
【Storage】Low temperature(subzero 15℃-subzero 20℃ ), sealed, lucifuge.
【Capacity】Raw material, 2kg/month. | 

